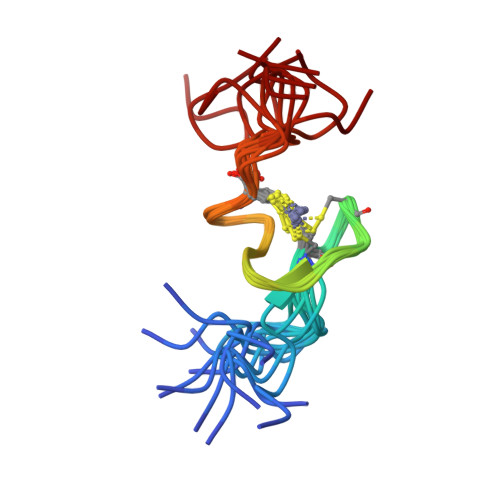
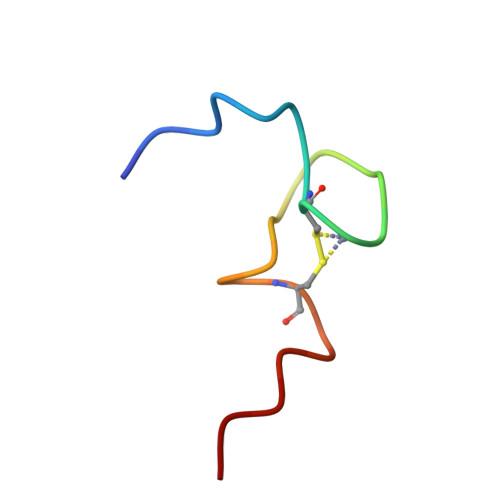
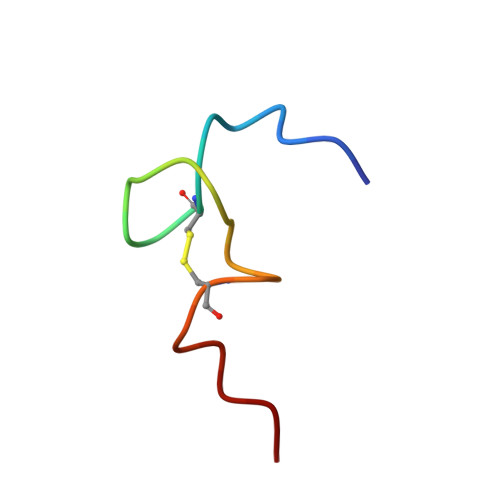
Structural role of the secondary active domain of HIV-2 NCp8 in multi-functionality
Matsui, T., Kodera, Y., Miyauchi, E., Tanaka, H., Endoh, H., Komatsu, H., Tanaka, T., Kohno, T., Maeda, T.(2007) Biochem Biophys Res Commun 358: 673-678
- PubMed: 17511966
- DOI: https://doi.org/10.1016/j.bbrc.2007.04.141
- Primary Citation of Related Structures:
2E1X - PubMed Abstract:
Nucleocapsid protein of HIV, containing two CCHC-type zinc fingers connected by a linker, is a multi-functional protein involved in many critical steps of the HIV life cycle. Several in vitro investigations demonstrated that the reactivities of the first zinc finger flanked by the linker of HIV-1 NCp7 and HIV-2 NCp8 were essential for binding to viral RNA, however, that of the second zinc finger flanked by the linker of NCp7 was very weak and non-specific, whereas the part of NCp8 called NCp8-f2, interacted strongly and specifically with viral RNA. In this study, the three-dimensional structure of NCp8-f2 was determined for the first time. Furthermore, we established that NCp8-f2 specifically binds to the stem-loop SD in viral RNA, and that the hydrophobic cleft and the basic residues close to the cleft were essential for specific binding to SD. We discuss the functional significance of NCp8-f2 for NCp8 being a multi-functional protein.
Organizational Affiliation:
Department of Physics, School of Science, Kitasato University, 1-15-1 Kitasato, Sagamihara, Kanagawa 228-8555, Japan.