Structure and mechanism of the lantibiotic cyclase involved in nisin biosynthesis
Li, B., Yu, J.P.J., Brunzelle, J.S., Moll, G.N., Van der Donk, W.A., Nair, S.K.(2006) Science 311: 1464-1467
- PubMed: 16527981
- DOI: https://doi.org/10.1126/science.1121422
- Primary Citation of Related Structures:
2G02, 2G0D - PubMed Abstract:
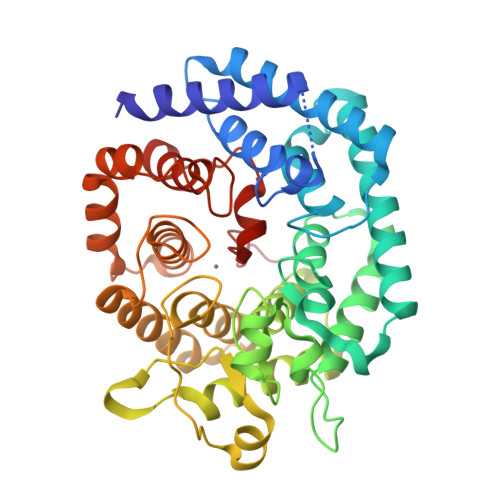
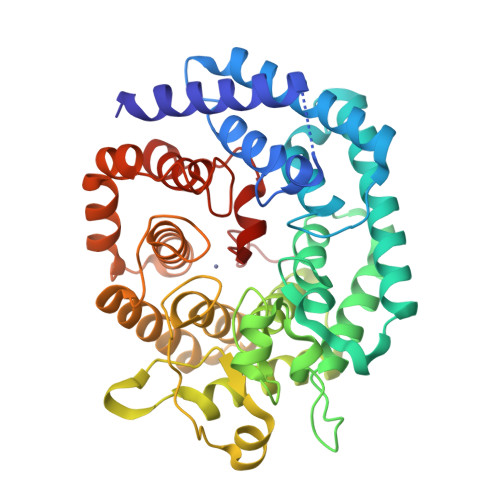
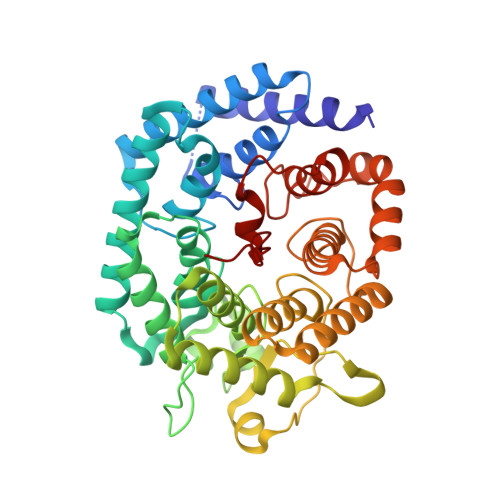
Nisin is a posttranslationally modified antimicrobial peptide that is widely used as a food preservative. It contains five cyclic thioethers of varying sizes that are installed by a single enzyme, NisC. Reported here are the in vitro reconstitution of the cyclization process and the x-ray crystal structure of the NisC enzyme. The structure reveals similarities in fold and substrate activation with mammalian farnesyl transferases, suggesting that human homologs of NisC posttranslationally modify a cysteine of a protein substrate.
Organizational Affiliation:
Department of Biochemistry, University of Illinois at Urbana-Champaign, 600 South Mathews Avenue, Urbana, IL 61801, USA.