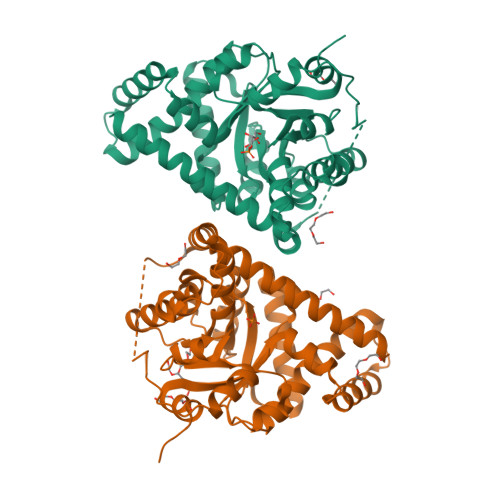
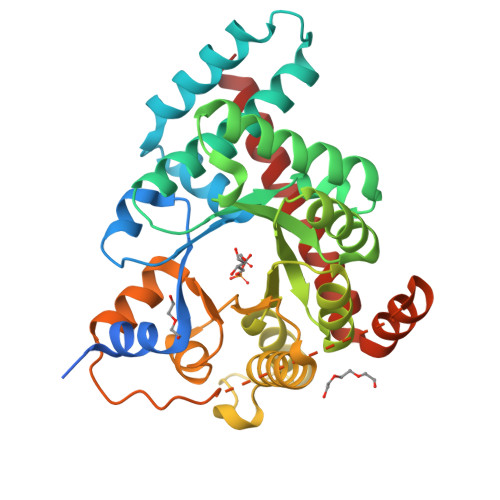
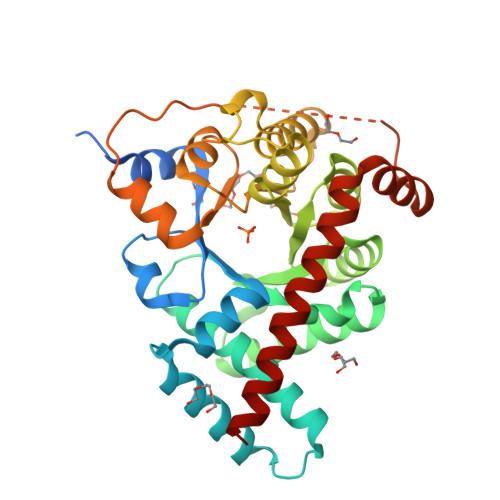
Adherence to Burgi-Dunitz stereochemical principles requires significant structural rearrangements in Schiff-base formation: insights from transaldolase complexes.
Light, S.H., Minasov, G., Duban, M.E., Anderson, W.F.(2014) Acta Crystallogr D Biol Crystallogr 70: 544-552
- PubMed: 24531488
- DOI: https://doi.org/10.1107/S1399004713030666
- Primary Citation of Related Structures:
3TE9, 3TK7, 3TKF, 3TNO, 4E0C - PubMed Abstract:
The Bürgi-Dunitz angle (αBD) describes the trajectory of approach of a nucleophile to an electrophile. The adoption of a stereoelectronically favorable αBD can necessitate significant reactive-group repositioning over the course of bond formation. In the context of enzyme catalysis, interactions with the protein constrain substrate rotation, which could necessitate structural transformations during bond formation. To probe this theoretical framework vis-à-vis biocatalysis, Schiff-base formation was analysed in Francisella tularensis transaldolase (TAL). Crystal structures of wild-type and Lys→Met mutant TAL in covalent and noncovalent complexes with fructose 6-phosphate and sedoheptulose 7-phosphate clarify the mechanism of catalysis and reveal that substrate keto moieties undergo significant conformational changes during Schiff-base formation. Structural changes compelled by the trajectory considerations discussed here bear relevance to bond formation in a variety of constrained enzymic/engineered systems and can inform the design of covalent therapeutics.
Organizational Affiliation:
Center for Structural Genomics of Infectious Diseases, USA.