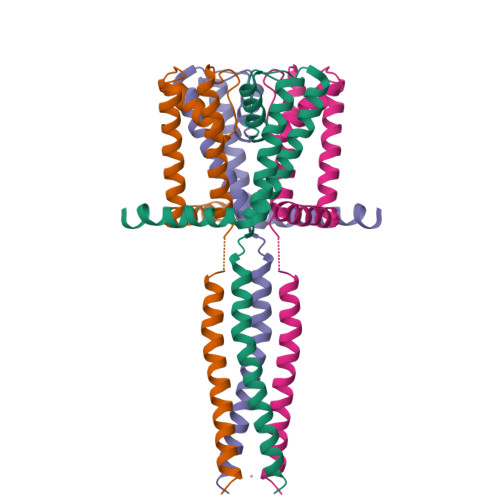
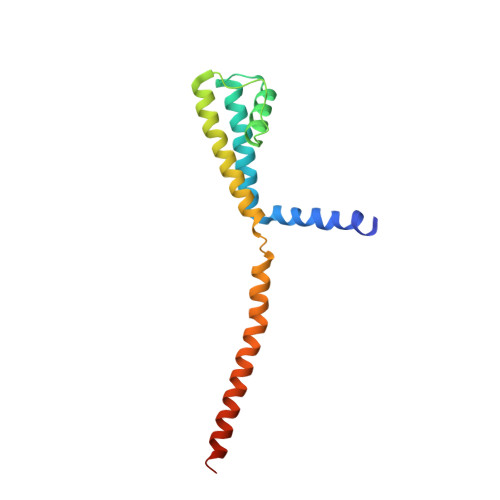
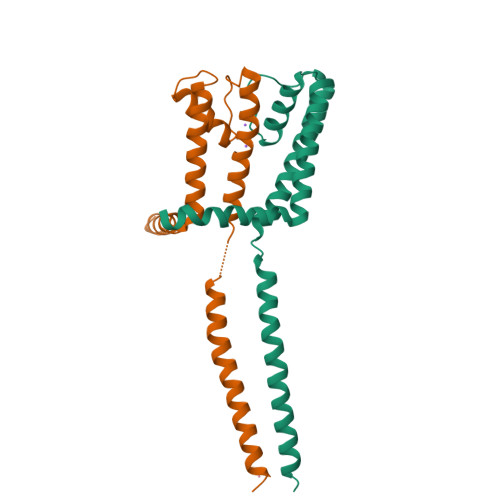The C-terminal helical bundle of the tetrameric prokaryotic sodium channel accelerates the inactivation rate
Irie, K., Shimomura, T., Fujiyoshi, Y.(2012) Nat Commun 3: 793-793
- PubMed: 22531178
- DOI: https://doi.org/10.1038/ncomms1797
- Primary Citation of Related Structures:
3VOU - PubMed Abstract:
Most tetrameric channels have cytosolic domains to regulate their functions, including channel inactivation. Here we show that the cytosolic C-terminal region of NavSulP, a prokaryotic voltage-gated sodium channel cloned from Sulfitobacter pontiacus, accelerates channel inactivation. The crystal structure of the C-terminal region of NavSulP grafted into the C-terminus of a NaK channel revealed that the NavSulP C-terminal region forms a four-helix bundle. Point mutations of the residues involved in the intersubunit interactions of the four-helix bundle destabilized the tetramer of the channel and reduced the inactivation rate. The four-helix bundle was directly connected to the inner helix of the pore domain, and a mutation increasing the rigidity of the inner helix also reduced the inactivation rate. These findings suggest that the NavSulP four-helix bundle has important roles not only in stabilizing the tetramer, but also in accelerating the inactivation rate, through promotion of the conformational change of the inner helix.
Organizational Affiliation:
Department of Biophysics, Graduate School of Science, Kyoto University, Oiwake, Kitashirakawa, Sakyo-ku, Kyoto 606-8502, Japan.