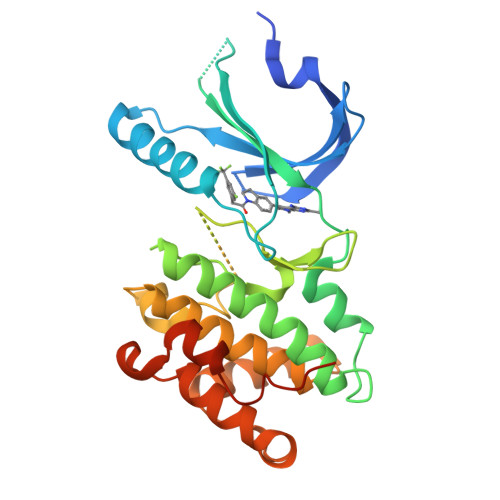
Discovery of 1H-Pyrazol-3(2H)-ones as Potent and Selective Inhibitors of Protein Kinase R-like Endoplasmic Reticulum Kinase (PERK).
Smith, A.L., Andrews, K.L., Beckmann, H., Bellon, S.F., Beltran, P.J., Booker, S., Chen, H., Chung, Y.A., D'Angelo, N.D., Dao, J., Dellamaggiore, K.R., Jaeckel, P., Kendall, R., Labitzke, K., Long, A.M., Materna-Reichelt, S., Mitchell, P., Norman, M.H., Powers, D., Rose, M., Shaffer, P.L., Wu, M.M., Lipford, J.R.(2015) J Med Chem 58: 1426-1441
- PubMed: 25587754
- DOI: https://doi.org/10.1021/jm5017494
- Primary Citation of Related Structures:
4X7H, 4X7J, 4X7K, 4X7L, 4X7N, 4X7O - PubMed Abstract:
The structure-based design and optimization of a novel series of selective PERK inhibitors are described resulting in the identification of 44 as a potent, highly selective, and orally active tool compound suitable for PERK pathway biology exploration both in vitro and in vivo.
Organizational Affiliation:
Departments of †Medicinal Chemistry, ‡Molecular Structure and Characterization, §Oncology Research, and ∥Pharmacokinetics and Drug Metabolism, Amgen Inc. , One Amgen Center Drive, Thousand Oaks, California 91320-1799, United States.