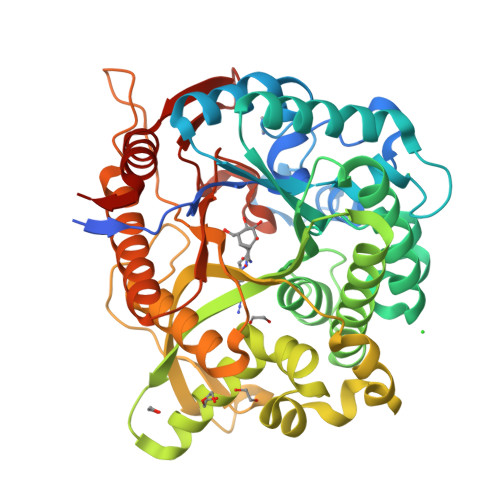
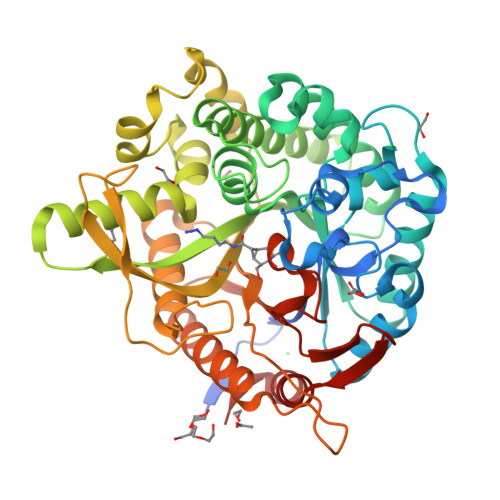
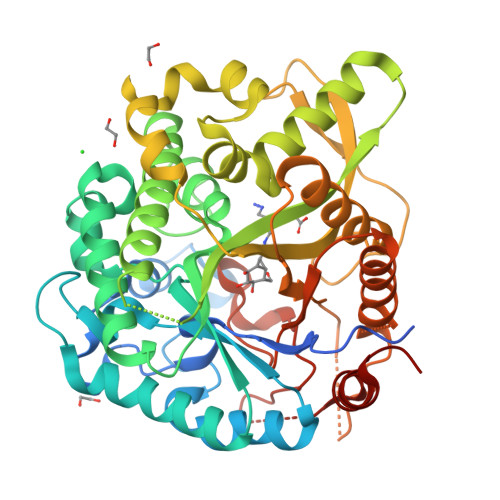
Carba-cyclophellitols Are Neutral Retaining-Glucosidase Inhibitors.
Beenakker, T.J.M., Wander, D.P.A., Offen, W.A., Artola, M., Raich, L., Ferraz, M.J., Li, K.Y., Houben, J.H.P.M., van Rijssel, E.R., Hansen, T., van der Marel, G.A., Codee, J.D.C., Aerts, J.M.F.G., Rovira, C., Davies, G.J., Overkleeft, H.S.(2017) J Am Chem Soc 139: 6534-6537
- PubMed: 28463498
- DOI: https://doi.org/10.1021/jacs.7b01773
- Primary Citation of Related Structures:
5N6S, 5N6T - PubMed Abstract:
The conformational analysis of glycosidases affords a route to their specific inhibition through transition-state mimicry. Inspired by the rapid reaction rates of cyclophellitol and cyclophellitol aziridine-both covalent retaining β-glucosidase inhibitors-we postulated that the corresponding carba "cyclopropyl" analogue would be a potent retaining β-glucosidase inhibitor for those enzymes reacting through the 4 H 3 transition-state conformation. Ab initio metadynamics simulations of the conformational free energy landscape for the cyclopropyl inhibitors show a strong bias for the 4 H 3 conformation, and carba-cyclophellitol, with an N-(4-azidobutyl)carboxamide moiety, proved to be a potent inhibitor (K i = 8.2 nM) of the Thermotoga maritima TmGH1 β-glucosidase. 3-D structural analysis and comparison with unreacted epoxides show that this compound indeed binds in the 4 H 3 conformation, suggesting that conformational strain induced through a cyclopropyl unit may add to the armory of tight-binding inhibitor designs.
Organizational Affiliation:
Department of Chemistry, University of York , Heslington, York, YO10 5DD, U.K.