Crystal structure of human GRP78 in complex with 8-aminoadenosine
Ferrie, R.P., Chen, Y., Antoshchenko, T., Cooney, O.M., Huang, Y., Park, H.W.To be published.
Experimental Data Snapshot
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Endoplasmic reticulum chaperone BiP | 382 | Homo sapiens | Mutation(s): 0 Gene Names: HSPA5, GRP78 EC: 3.6.4.10 | 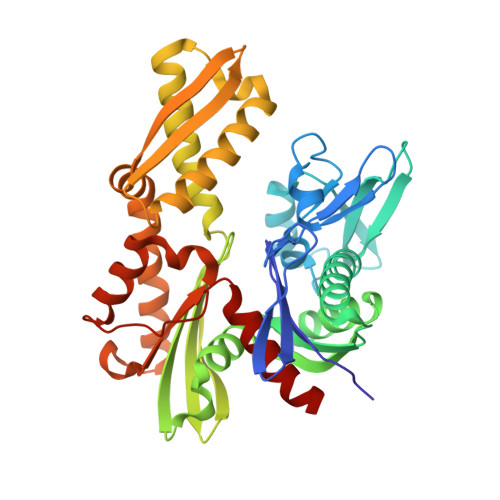 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P11021 (Homo sapiens) Explore P11021 Go to UniProtKB: P11021 | |||||
PHAROS: P11021 GTEx: ENSG00000044574 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P11021 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 3BH Query on 3BH | C [auth A], D [auth B] | (2R,3R,4S,5R)-2-(6,8-diaminopurin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol C10 H14 N6 O4 DVGWFQILDUEEGX-UUOKFMHZSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 55.662 | α = 90 |
| b = 74.775 | β = 99.48 |
| c = 89.485 | γ = 90 |
| Software Name | Purpose |
|---|---|
| XDS | data reduction |
| SCALA | data scaling |
| MOLREP | phasing |
| BUSTER | refinement |
| PDB_EXTRACT | data extraction |