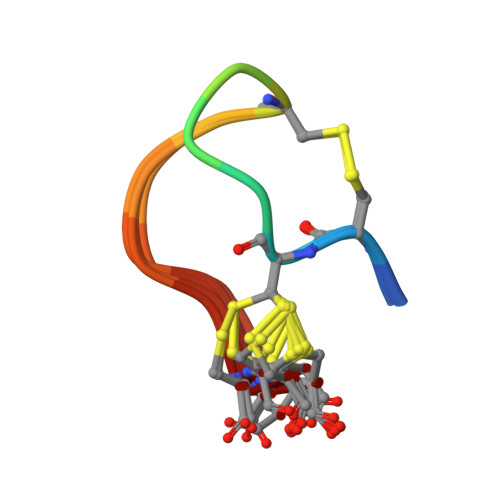
Cysteine-Rich alpha-Conotoxin SII Displays Novel Interactions at the Muscle Nicotinic Acetylcholine Receptor.
Wilhelm, P., Luna-Ramirez, K., Chin, Y.K., Dekan, Z., Abraham, N., Tae, H.S., Chow, C.Y., Eagles, D.A., King, G.F., Lewis, R.J., Adams, D.J., Alewood, P.F.(2022) ACS Chem Neurosci 13: 1245-1250
- PubMed: 35357806
- DOI: https://doi.org/10.1021/acschemneuro.1c00857
- Primary Citation of Related Structures:
6OVJ - PubMed Abstract:
α-Conotoxins that target muscle nicotinic acetylcholine receptors (nAChRs) commonly fall into two structural classes, frameworks I and II containing two and three disulfide bonds, respectively. Conotoxin SII is the sole member of the cysteine-rich framework II with ill-defined interactions at the nAChRs. Following directed synthesis of α-SII, NMR analysis revealed a well-defined structure containing a 3 10 -helix frequently employed by framework I α-conotoxins; α-SII acted at the muscle nAChR with half-maximal inhibitory concentrations (IC 50 ) of 120 nM (adult) and 370 nM (fetal) though weakly at neuronal nAChRs. Truncation of α-SII to a two disulfide bond amidated peptide with framework I disulfide connectivity led to similar activity. Surprisingly, the more constrained α-SII was less stable under mild reducing conditions and displayed a unique docking mode at the nAChR.
Organizational Affiliation:
Institute for Molecular Bioscience, The University of Queensland, St Lucia, QLD 4072, Australia.